Introduction: The Foundation of Every Bar
At first glance, a paint manufacturing floor and an artisanal soap studio might seem worlds apart. However, as a company with 80 years of experience in industrial chemistry, we know that the heart of both crafts lies in the same place: the precise control of chemical reactions.
Mastering sodium hydroxide in soap making is the bridge between being a hobbyist and a professional maker. Whether you call it caustic soda for soap or lye for soap making, understanding this ingredient is essential for safety and quality.
In this guide, we are bridging the gap between industrial-grade knowledge and handcrafted artistry to show you how to master sodium hydroxide in soap making with the confidence of a professional chemist.
What is Sodium Hydroxide? Understanding the “Lye”
In the soap-making community, you will hear three terms used interchangeably. While they refer to the same active ingredient, understanding the “why” behind the names helps you navigate supplier catalogues and safety data sheets (SDS).
Understanding the Industry Terms
Sodium Hydroxide (NaOH)
This is the scientific name and the formal chemical identity. It consists of one sodium atom, one oxygen atom, and one hydrogen atom. In technical or older manufacturing contexts, you might see it referred to as “Sodium Hydrate.” When you are looking for the purest possible ingredient for your soap, this is the name you should look for on the label.
Caustic Soda
This is the descriptive name. It highlights the chemical’s physical nature.
Caustic: Derived from the Greek word kaustikos, meaning “able to burn.” This serves as a primary warning that the substance is corrosive to organic tissue (like skin and eyes).
Soda: Historically, “soda” was a catch-all term for various sodium salts.
Lye
“Lye” is the common or traditional name. Historically, soap makers did not use lye as a solid pellet but a liquid solution created by leaching water through hardwood ashes. This ash-water (mostly potassium hydroxide) was used for centuries to make soft soaps. Today, in modern cold-process soap making, “Lye” is the term we use for the solution created when we dissolve pure Sodium Hydroxide beads into water.
💡Pro-Tip: Sodium Hydroxide is hygroscopic (it attracts water from the air), it must always be stored in airtight, non-metallic containers.
The Chemistry: How Soap is Actually Made
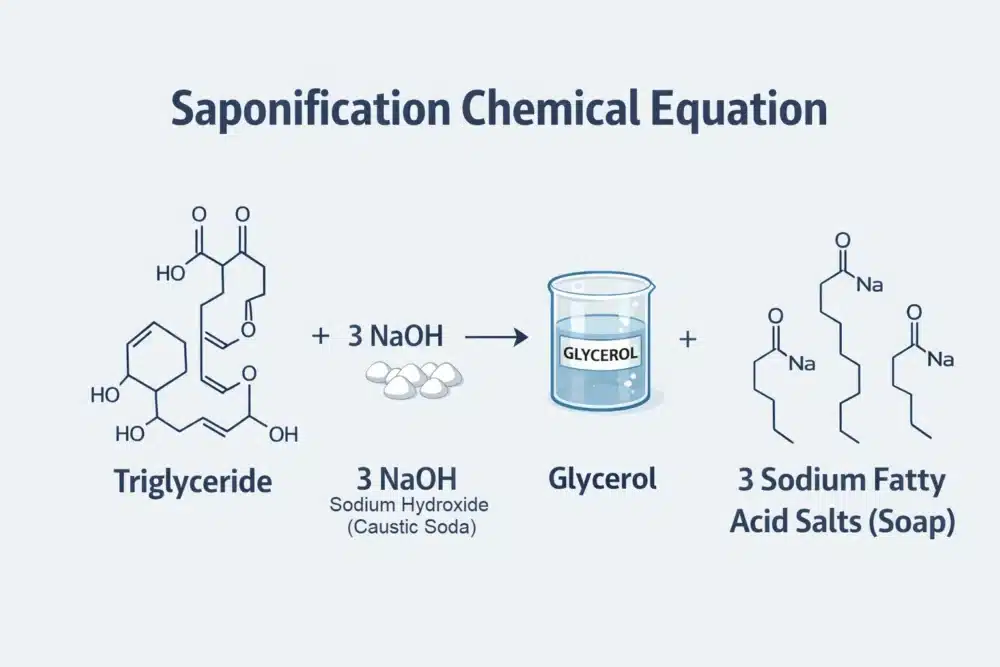
What is Saponification?
Saponification is the chemical reaction between a fat or oil (triglyceride) and a strong alkali such as sodium hydroxide (caustic soda) that produces soap and glycerol.
- The Chemist’s Note: While sodium hydroxide is used to create bar soap, its cousin—Potassium Hydroxide (KOH)—is used for liquid soap. Both are types of lye, but they are not interchangeable in recipes.
To the uninitiated, “lye” sounds like a harsh additive. To a chemist, it is a vital reagent. You cannot have “true” soap without a specific chemical reaction.
How Oils Become Soap
The saponification process explained simply is a reaction between an acid (oils/fats) and a base (sodium hydroxide). When these two meet in the presence of water, they undergo a molecular divorce and remarriage. The triglycerides in the fat break apart, and the fatty acids bond with the sodium ions to create a completely new substance: soap
-
The Byproduct: The reaction naturally creates glycerol. In industrial soap, this is often removed; in handmade soap, it remains to moisturize the skin.
-
The Result: A salt of a fatty acid—what we call a soap bar.
Essential Safety: Sodium Hydroxide Safety Handling
With 80 years in the chemical industry, safety is our second language. Sodium hydroxide is a “Strong Base,” meaning it can cause chemical burns if it touches the skin or eyes. However, it is not “scary” if you respect it.
Essential PPE (Personal Protective Equipment)
-
Safety Goggles: Not just glasses. You need wrap-around protection to prevent splashes.
-
Chemical-Resistant Gloves: Nitrile or neoprene are preferred over thin latex.
-
Long Sleeves and Pants: Cover all exposed skin
-
Proper Ventilation: Mixing lye with water creates brief but potent fumes. Always mix under a vent hood or near an open window.
The “Lye into Water” Rule
Never pour water into lye. This can cause a “lye volcano”—a sudden, violent eruption of caustic heat.
-
Always add the Sodium Hydroxide TO the water. * Stir gently and continuously until dissolved.
Equipment Science: Choosing the Right Containers
Sodium hydroxide is highly reactive. Using the wrong equipment can lead to container failure or contaminated soap.
-
The “No-Go” List: Aluminum, Tin, and Zinc. These metals react with lye to produce flammable hydrogen gas. Never use a “silver” pot unless it is high-quality Stainless Steel.
-
The “Safe” List: Stainless Steel (304 or 316 grade) and HDPE #2 Plastic. Most heavy-duty mixing bowls and “tupperware” are made of HDPE #2, which is heat-resistant and chemically stable.
Formulating Your Recipe: Lye Calculations
A successful batch of soap depends on math. You cannot “eyeball” the amount of lye you use.
What is a SAP Value?
Every oil has a Saponification (SAP) Value. This number represents the exact amount of Sodium Hydroxide required to turn 1 gram of that specific oil into soap.
- Coconut Oil has a high SAP value (it needs more lye).
- Olive Oil has a lower SAP value (it needs less lye).
The Concept of “Superfatting”
To ensure safety and skin comfort, most soap makers use a 5% Lye Discount, also known as “Superfatting.”
By using 5% less lye than the math requires, you ensure:
1. There is no chance of “active lye” remaining in the bar.
2. There is extra oil left over to provide a moisturizing, luxurious feel to the skin.
Purity Matters: Choosing Your Caustic Soda for Soap
As a chemical manufacturer, we emphasize purity. Many “hardware store” lyes contain anti-clumping agents or drain cleaners. For soap making, you must use Food Grade or Technical Grade Sodium Hydroxide (98%+ purity). Impurities trigger unpredictable reactions, off-putting smells, or skin irritation. When sourcing caustic soda for soap, avoid the hardware store “drain cleaner” aisle. Those products often contain anti-clumping agents or aluminum bits that can ruin a batch of soap or cause dangerous gas. Always seek 98-99% pure Sodium Hydroxide (NaOH) specifically packaged for cosmetic or food use.
FAQ: Common Questions About Sodium Hydroxide
Common Soap Making Myths
Q: Can I make soap without sodium hydroxide?
A: No. All “real” soap is made with lye. “Melt and Pour” bases are often marketed as lye-free, but they were simply made with lye by a manufacturer before you purchased them.
Q: How do I dispose of leftover lye water?
A: Neutralize it by slowly adding it to a large bucket of water, then flush it down the drain with plenty of running water. Alternatively, use it as a heavy-duty drain cleaner—which is its original industrial use!
Q: Is lye soap “natural”?
A: Yes. While sodium hydroxide is a processed chemical, it is completely consumed during the saponification process. No lye remains in a properly made, cured bar of soap.
Disposal and Storage
Q: How long should I store sodium hydroxide?
A: If kept in a dry, airtight container, it has a shelf life of about 1 year. If it becomes clumpy or “wet,” its potency has decreased, and your lye calculations may be off.
Ready to start your soap making journey with professional-grade precision?
Whether you are a hobbyist or looking to start a business, understanding the chemistry of your ingredients is the first step toward quality.
Contacting us is easy!
Email: sales@palatinepaints.co.uk
Call Us: 01942 884 122
Contact form: https://www.palatinepaints.co.uk/contact-us
As always, we welcome comments below. We also have our online chat manned each weekday during working hours or call/email.