POSTED IN: HYGIENE | READING TIME: 6 MINUTES | LAST UPDATED: 6TH MARCH 2026
Sodium hydroxide bridges the gap between raw oils and finished soap bars by driving the essential saponification reaction. This guide delivers industrial-grade expertise on handling Caustic Soda safely while mastering the complex chemistry of lye. You will learn how to produce high-quality soap with the precision and confidence of a professional chemist.
Understanding the Industry Terms: NaOH, Caustic Soda, and Lye
Soap makers often use three different names for the same active ingredient. Sodium Hydroxide (NaOH) represents the formal scientific identity, which consists of sodium, oxygen, and hydrogen atoms. However, suppliers frequently use the terms Caustic Soda or Lye in their catalogues. Consequently, you must recognise these synonyms to navigate safety data sheets effectively.
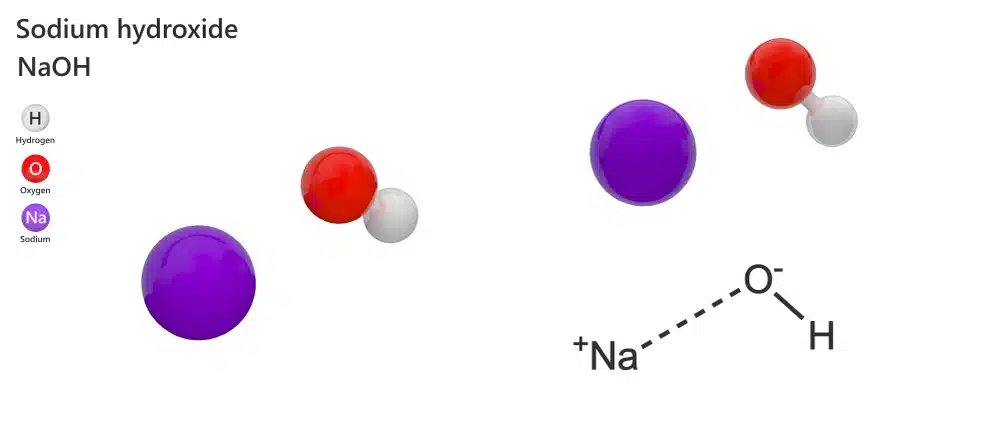
The Chemical Structure of Caustic Soda.
The name “Caustic Soda” specifically describes the chemical’s physical nature because the word “caustic” refers to its ability to burn organic tissue. Conversely, “Lye” serves as the traditional common name for the liquid solution. Therefore, understanding these labels helps you source the purest ingredients for your craft.
Furthermore, you should distinguish Sodium Hydroxide from Potassium Hydroxide (KOH). While both act as types of lye, you only use Sodium Hydroxide for solid bar soaps. On the other hand, liquid soap recipes require Potassium Hydroxide. As a result, using the wrong chemical will inevitably ruin your batch.
The Chemistry of Saponification
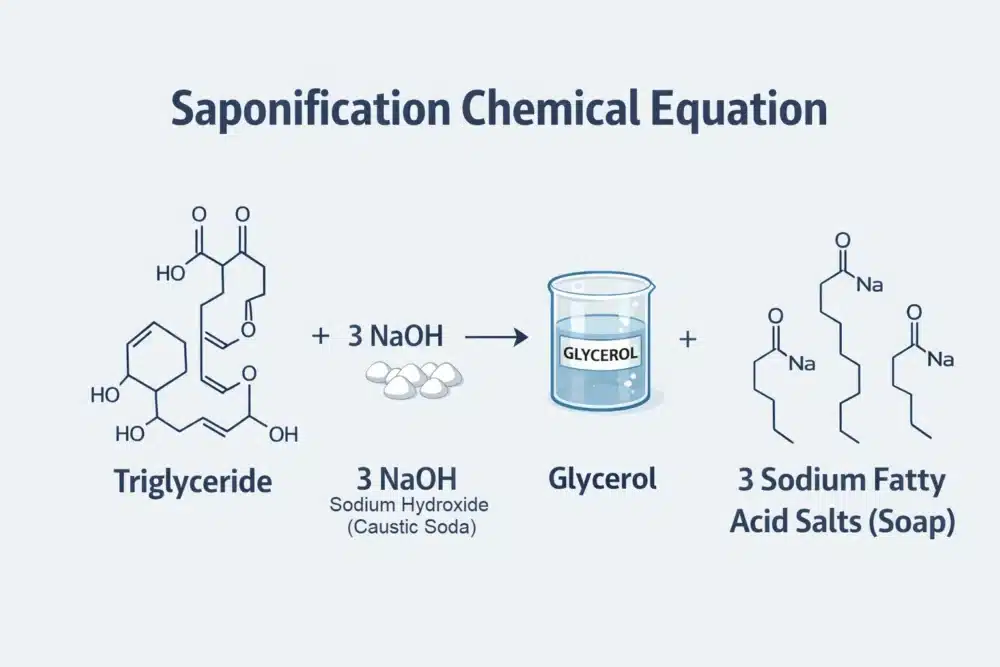
Saponification represents the chemical heart of soap making. In simple terms, this reaction occurs between an acid and a base. When these two meet in water, they undergo a molecular transformation. The triglycerides in the oil break apart. Subsequently, the fatty acids bond with sodium ions to create an entirely new substance.
This process naturally generates glycerol as a useful byproduct. Industrial manufacturers often remove this glycerin for separate sale. However, handcrafted soap retains this glycerol to moisturise the skin. Consequently, handmade bars usually feel much gentler than mass-produced soaps. As a result, the final product offers superior skin benefits.
Eventually, the saponification reaction consumes all the sodium hydroxide. If you calculate your recipe correctly, no lye remains in the finished bar. Therefore, the corrosive chemical disappears entirely during the curing process. For this reason, we recommend using our high-purity caustic soda to ensure perfect results every time.
Saponification: A Molecular Transformation
| Initial Ingredients | Saponification Reaction | Resulting Products |
| Triglycerides (Oils/Fats) |
Molecules break apart. |
Soap (Sodium Salts). |
| Caustic Soda (Lye) |
Caustic soda is consumed. |
Glycerin (Glycerol) |
| Water |
Promotes reaction; dries off. |
Cured Bar (Dry & Mild) |
Key Takeaway: Saponification is a complete chemical transformation; when calculated correctly, the corrosive sodium hydroxide and the greasy oils are entirely replaced by soap and moisturising glycerol.
High-purity sodium hydroxide pellets, often referred to as caustic soda or lye in soap making.
Essential Safety and PPE Requirements
Safety must remain your top priority when handling sodium hydroxide. Because this chemical acts as a strong base, it can cause severe burns on contact. Therefore, you must wear full Personal Protective Equipment (PPE) at all times. Specifically, choose wrap-around goggles instead of standard glasses to block accidental splashes.
In addition, wear chemical-resistant gloves and long-sleeved clothing to protect your skin. On the other hand, if a splash occurs, immediately rinse the area with cool water. Furthermore, never mix lye with other chemicals like ammonia. Doing so can release toxic chlorine gas. Consequently, you must always work in a well-ventilated space.
Mixing Safety: The “Snow on the Lake” Rule
You must follow a specific sequence when mixing your lye solution. Always add the lye pellets into the water. Never pour water into a container of lye. Otherwise, the mixture may erupt or “volcano” out of the jug. Therefore, remember the phrase “snow falls on the lake” to keep your process safe.
Storage and Shelf Life Best Practices
Sodium hydroxide is highly hygroscopic, meaning it aggressively attracts moisture from the air. If you leave a container open, the pellets will begin to clump together and turn into a “wet” slush. As a result, the chemical becomes less potent by weight, which will throw off your precise soap recipes and potentially lead to an oily, “soft” batch of soap.
To prevent this, always store your Caustic Soda in an airtight, non-metallic container (high-density polyethylene or HDPE is preferred). If kept in a dry, cool environment, sodium hydroxide typically has a shelf life of about one year. However, if your pellets look clumpy or feel damp, it is a sign that they have absorbed moisture and should be replaced for consistent results.
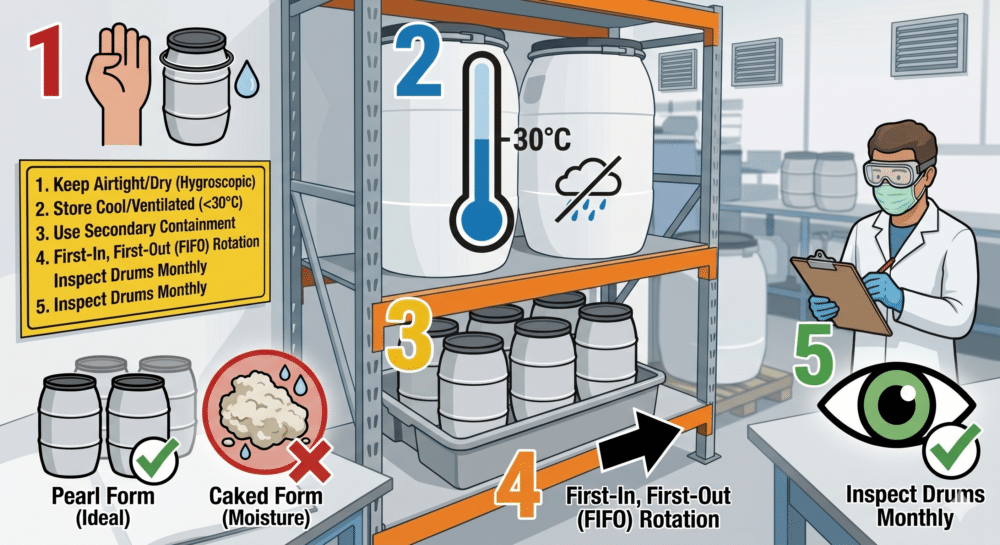
How to Correctly Store Caustic Soda
Important: Lye reacts aggressively with metals like aluminium and zinc. Therefore, you must only use stainless steel or heat-resistant plastic tools when mixing your lye solution.
Neutralisation and Disposal Procedures
After your soap-making session, you must dispose of any leftover materials safely. First, neutralise your lye water by diluting it in a large bucket of plain water. Then, you can safely flush the solution down the drain. Furthermore, run plenty of cold water afterwards to clear the pipes effectively.
In fact, many industries use sodium hydroxide as a powerful drain cleaner. Consequently, flushing a diluted solution often benefits your plumbing. On the other hand, you should contact local hazardous waste facilities for large-scale disposal of dry pellets. Thus, following these steps protects both your home and the environment.
Now that you understand these industrial standards, you can craft soap with professional skill. By following these safety and chemical guidelines, you ensure that every bar meets the highest quality levels. Consequently, your knowledge of reagents will elevate your artisanal soap business or hobby to a new standard.
Ready to get started?
Browse our full range of Caustic Soda – available for next-day delivery or in-store collection from Leigh, Greater Manchester.
Frequently Asked Questions
Jason | Technical Director at Palatine Paints
With over 30 years in the coatings industry, Jason regularly advises trade and industrial customers on surface preparation and paint systems. Have a question about this guide? Get in touch.